Terbium-161 Separation
 The beta emitting isotope 161Tb has garnered interest in nuclear medicine for targeted radionuclide therapy.[1,2] 161Tb is produced by irradiation of Gd targets.
The beta emitting isotope 161Tb has garnered interest in nuclear medicine for targeted radionuclide therapy.[1,2] 161Tb is produced by irradiation of Gd targets.
Due to the small separation factors between adjacent rare earth elements, the separation of 161Tb from 10-300 mg of Gd target material requires relatively long columns, small particle size resin, elevated temperature and multiple separation columns to achieve the desired high specific activity final products.[1,3,4] Two main chromatographic systems have been applied to the separation of adjacent rare earths: 1) Extraction chromatographic resins (EXC) based on organophoshorus extractants (HDEHP = LN Resin and HEH[EHP] = LN2 Resin) from mineral acids (HNO3 and HCl) and 2) Strong acid cation exchange resin (50Wx8 or MP50) from alpha hydroxybutyric acid (a-HIBA).[1]
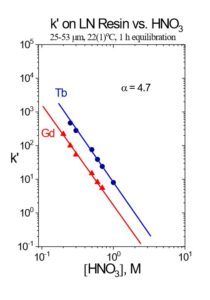
In the EXC separation schemes, LN2 or LN resin is used to perform the bulk of the Tb/Gd separation. The separation factors for adjacent rare earth elements is normally small, ranging from 1.5 to 4.9.[5] The separation factor is 4.7 for Tb/Gd on LN Resin (Figure 1). Separation factors for LN and LN2 resins are normally very similar, and heavier (higher atomic number) rare earth metal ions are retained more strongly than lighter (lower atomic number) rare earth metal ions. The less acidic HEH[EHP] extractant on LN2 allows recovery of rare earths in more dilute acid than the more acidic HDEHP on LN resin. Therefore, LN2 resin is often used for separation of heavier rare earth elements (Dy-Lu), Y and Sc, while LN is more often used for lighter rare earth elements (La-Tb), and Ac. When the loading of LN or LN2 resin is low (<5% functional capacity) adjacent rare earth elements can be separated efficiently using a single column of f-grade (25-50 mm) resin (Figure 2).[3,5] (column = 25-50 μm resin, diameter = 1.0 cm, length = 13 cm, column volume 10 mL).
With relatively large targets 50-300 mg of Yb or Gd, it may be impractical to pack a single column large enough to keep the functional capacity below 5%. Furthermore, a single large column may not provide adequate decontamination of the desired radionuclide from the target material to achieve the high specific activities and “no carrier added” (n.c.a) designation required for efficient labeling of 161Tb to biolocalization agents for targeted radionuclide therapy. In these cases, a series of 2-3 LN resin columns, operated at higher capacity, can provide a more efficient separation.[1,3]
Fe(III) may co-elute with Tb and must be separated in subsequent steps using DGA, normal resin.
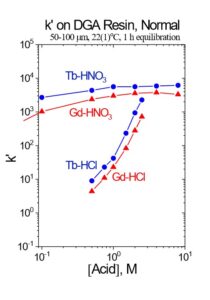
DGA resin, normal may be used between successive LN columns to concentrate the Tb and convert from the relatively high concentration of HNO3 used to elute the LN and the low concentration of HCl required to load the subsequent LN column. Utilizing the DGA resin, normal eliminates the need for evaporations, enables the rapid separation of 161Tb using up to 3 LN column separations, and helps to remove reagent impurities, such as Al, Ca, and Fe.[3,6] The Tb fraction from the first LN column is loaded directly onto a column of DGA resin, normal, rinsed with 0.10 M HNO3 to reduce the acidity, and then Tb is recovered using 0.05M HCl (Figure 4). The Tb in 0.05M HCl is then loaded onto the second LN resin column which further reduces the Gd content, while recovering the Tb in high yield.
As long as there is a significant mass of Gd (>0.2 mg) remaining with the 161Tb fraction, recovery of 161Tb from the DGA, resin, normal with dilute HCl is very efficient. However, after the second LN separation, when nearly all of the Gd has been removed, the recovery of 161Tb from the DGA resin can be retarded by the small amount of the HDEHP extractant that has leached from the LN resin.[3] The acidic HDEHP extractant leached from LN is adsorbed onto the DGA resin and complexes 161Tb, leading to a broad elution band and lower recovery from dilute HCl. The impact of the LN extractant leach is minimized by introducing a small guard cartridge of UTEVA resin above the final DGA Resin column. The UTEVA resin adsorbs the HDEHP and is removed prior to stripping the 161Tb from DGA, leading to more complete recovery of 161Tb in a small volume of dilute HCl. Passing the final 161Tb eluate from the DGA resin column through a small column of anion exchange resin in the chloride form (A8-F-CL, strong base anion exchange) helps to remove any residual nitrate.
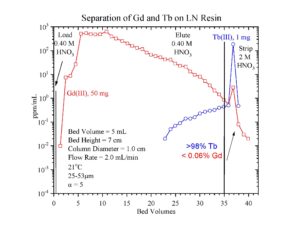
The separation of 161Tb from Gd targets is simpler than the separation of 177Lu from Yb targets due to the much larger separation factor between Tb and Gd (a = 4.7 on LN Resin) than Lu and Yb (a = 1.7 on LN2 Resin). For 161Tb, a single LN column operated at ~80% of functional capacity at room temperature can achieve >98% recovery of 161Tb while removing >99.5% of the Gd target (Figure 4). Following concentration of the 161Tb on DGA Resin, Normal, a relatively small second column of LN2 resin then provides additional decontamination from Gd. The 161Tb is then concentrated on a second small column of DGA Resin, Normal (with a UTEVA guard column to remove any HDEHP leached from the LN columns) and recovered in dilute HCl. The analogous separation of 177Lu from irradiated Yb targets requires 3 relatively large LN2 columns.
References
[1] Dash, A., Raghavan, M., Pillai, A. Knapp, F.F. 2015. Production of 177Lu for Radionuclide Therapy: Available Options, Nucl. Med. Mol. Imaging, 49, 85-107.
[2] Lehenberger, S., Barkhausen, C., Cohrs, S., Fischer, S., Grunberg, J., Hohn, A., Koster, U., Schibli, R., Turler, A., Zhernosekov, K., 2011. The low energy b- and electron emitter 161Tb as an alternative to 177Lu for targeted radionuclide therapy, Nucl. Med. Bio., 38, 917-924.
[3] Horwitz, E.P. McAlister, D.R. Bond, A.H. Barrans, Jr., R.E. Williamson, J.M. 2005. A Process for the Separation of 177Lu from Neutron Irradiated 176Yb Targets, Applied Radiation and Isotopes, 63, 23-36.
[4] Mirzadeh, S., Du, M., Beets, A. L., Knapp, Jr., F. F., April 6, 2004. Method for preparing high specific activity 177Lu. United States Patent No. 6,716,353.
[5] McAlister, D.R. Horwitz, E.P. 2007. The Characterization and Novel Applications of Extraction Chromatographic Materials Containing Bis(2-ethyl-1-hexyl)phosphoric Acid, 2-ethyl-1-hexylphosphonic acid, mono 2-ethyl-1-hexyl ester and 2,4,4-trimethyl-1-pentylphosphinic acid, Solv. Extr. Ion Exch., 25(6), 757-769.
[6] Horwitz, E.P. McAlister, D.R. Bond, A.H. Barrans, Jr., R.E. 2005. Novel Extraction Chromatographic Resins Based on Tetraalkyldiglycolamides: Characterization and Potential Applications,” Solv. Extr. Ion Exch., 23, 319-344 (2005).